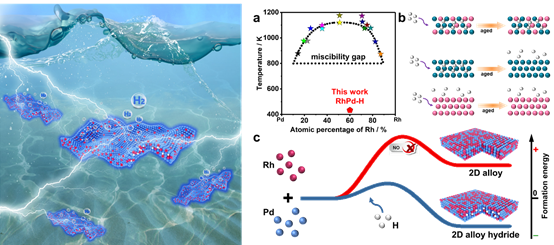
Hydrogen Stabilized RhPdH 2D Bimetallene Nanosheets for Efficient Alkaline Hydrogen Evolution
Recently, Prof. Xiaoqiang Cui from the Department of Materials Science of the Jilin University reported an efficient catalyst of two-dimensional bimetallene hydrides, achieving outstanding alkaline HER performances concurrently with high activity and robust stability. This work provides guidance for synthesizing high-activity catalysts by confining hetero-atoms into the crystal lattice of bimetallene and also a very novel mechanism for the growth of bimetallene made of highly immiscible components.
Hydrogen evolution reaction (HER) from water splitting is a fundamental process in the electrocatalysis and plays a crucial role for hydrogen-based energy sources. Industrial HER is normally under alkaline media conditions. However, due to the sluggish kinetics of the prior water dissolution step (H2O → H* + OH-), the reaction rate is the orders of magnitude lower than that in acid conditions. Unfortunately, most of the reported catalysts can operate only steadily at low current density (10 mA cm−2), which is far from the standards of commercial applications (at least 100 mA cm−2). Therefore, it is necessary to develop a catalyst possessing both high activity and robust stability for alkaline HER.
Here, Prof. Xiaoqiang Cui reported the development of a high-efficient stable electrocatalyst by confining H atoms to form RhPdH bimetallene hydride. The system exists because of the introduction of H that is in-situ chemically released from the formaldehyde solution during the synthesis. This provides a highly stable catalyst based on an unstable combination of metal elements. Density functional theory calculations show the H is confined by electronic interactions and the Miedema rule of reverse stability of the RhPd alloy. The obtained catalyst exhibits outstanding alkaline HER catalytic performance with a low overpotential of 40 mV at 10 mA cm-2 and remarkable stability for over 10 hours at 100 mA cm-2. The experimental results show that the confined H improve the activity, while the ultrathin sheet-like morphology yields stability.
This work has been published as an article in J. Am. Chem. Soc. This work was supported by the National Natural Science Foundation of China, the Program for JLU Science and Technology Innovative Research Team, and the Fundamental Research Funds for the Central Universities.
Link: https://pubs.acs.org/doi/abs/10.1021/jacs.0c00218
https://www.x-mol.com/news/447489